WHO WE ARE?
Prolistem Therapies is a biotechnology company originating from the U.S., with clinical experience linked to MD Anderson Cancer Center and global expertise in cell-based therapeutics. Our initiative focuses on producing CAR-T and future TIL, NK, and TCR therapies under full GMP conditions
OUR ROLE
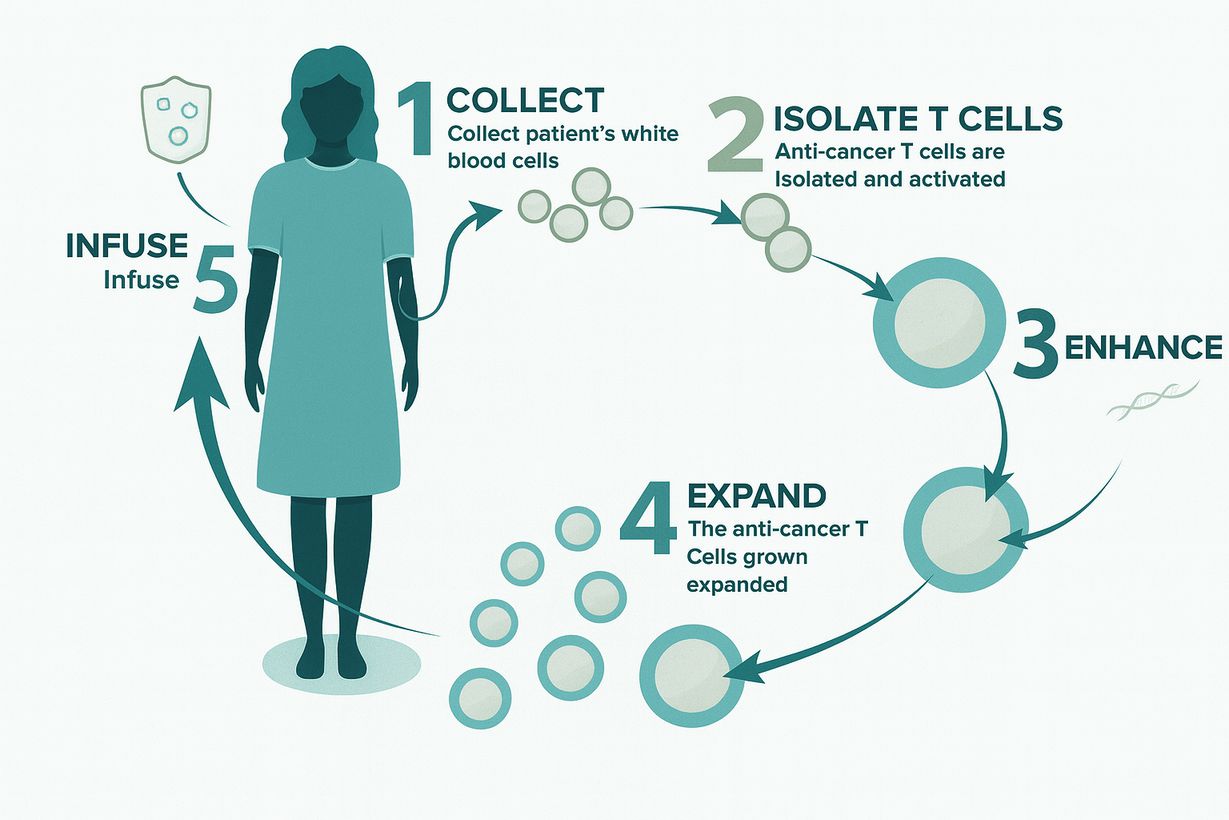
We handle the complete GMP manufacturing process of the
CAR-T cellular product. Partner university hospitals conduct
patient selection, pre-conditioning, infusion, and post-care.
Production takes place inside the university’s own medical
campus, eliminating the need for cryogenic freezing or
external transport
OUR ADVANTAGE
Through modular clean-room technology and process automation,
Prolistem enables hospitals to achieve full CAR-T readiness
without capital strain.
The model has already proven effective in U.S. and European
facilities aiming for decentralized manufacturing.
WHY PARTNER WITH US
Clinically Validated CAR-T Platform
Gain access to a clinically validated CAR-T manufacturing
platform with Comprehensive staff training and certification
10% CapEx vs. Conventional
No need for multi-million-dollar infrastructure, our solution
delivers the same output at only 10% of conventional
investment
GMP-Grade Operations
Compliance with international GMP standards (ISO Class 7
environment, validated SOPs, digital batch records)
Academic Partnership & Clinical Visibility
Joint academic visibility: co-authored papers, clinical data
access, and conference representation.
PATIENT PIPELINE COMMITMENT
Prolistem ensures clinical continuity: over the next two years, we can directly contribute a sufficient number
of patient cases to keep the laboratory at full utilization, protecting both research output and clinical
relevance.
COLLABORATION MODEL
- Production – Prolistem establishes and operates the GMP manufacturing lab within the university’s medical complex.
- Clinical Treatment – The university hospital performs patient preparation, infusion, and monitoring.
- Shared Outcomes – Both parties benefit from data generation, publications, and patient outcomes.

ECONOMIC HIGHLIGHTS
- Traditional CAR-T facility setup requires about USD 2 million+. Prolistem’s modular model reduces this by 90% without compromising GMP quality
- Total treatment goal: below USD 100,000 per patient, making it regionally accessible.
- Full installation, validation, and staff training are included in the collaboration.
- On-campus production eliminates freezing, shipment delays, and logistics costs.
STRATEGIC ADVANTAGES FOR UNIVERSITY HOSPITALS
- Become one of the first CAR-T capable centers in Northern Cyprus.
- An immediate on-site demonstration is available for quality and efficacy proof.
- Clinical and academic prestige through early adoption.
- Opportunity for international collaborations and joint publications.
- Knowledge transfer and skill development for postgraduate students.
- Sustainable expansion to TIL, NK, and TCR pipelines.
IMPLEMENTATION ROADMAP (IMMEDIATE START)
- Immediate: On-site demonstration and validation in existing university laboratories (quality and efficacy proof).
- Weeks 1–4: Laboratory establishment and GMP system setup.
- Week 8: First patient infusion under pilot protocol.
OUR TEAM
Prolistem Therapies is led by expert scientists with deep experience in cancer biology, stem cell research, and
cell-therapy manufacturing and automation—spanning the Technion–Israel Institute of Technology and the MD
Anderson Cancer Center (USA). The team developed a patented therapeutic approach and currently directs multiple
programs, including CAR-T, TC, R, NK, and TIL therapies. We have optimized hundreds of cell-therapy protocols in
collaboration with leading centers in the United States, Israel (Sheba & Hadassah), Germany,
Switzerland, and the Netherlands. Together, we’ve partnered with top institutions across the US, Europe, and the
Middle East to refine autologous and allogeneic workflows, align QC /QA systems, and improve manufacturability.
